
Sir Walter Raleigh. Wikipedia image.
Sir Walter Raleigh is reputed to have won a bet with Queen Elizabeth that he would be able to weigh the smoke coming from his pipe. After she accepted the bet, he weighed a pinch of tobacco, smoked it, and then weighed the resulting ashes. He convinced the Queen that the difference in weight was the weight of the smoke.
Of course, the Queen could have won the bet by pointing out that the combustion products contained oxygen, and most of that oxygen originated not in the tobacco, but in the air. But she didn’t think of that, and instead paid the bet.
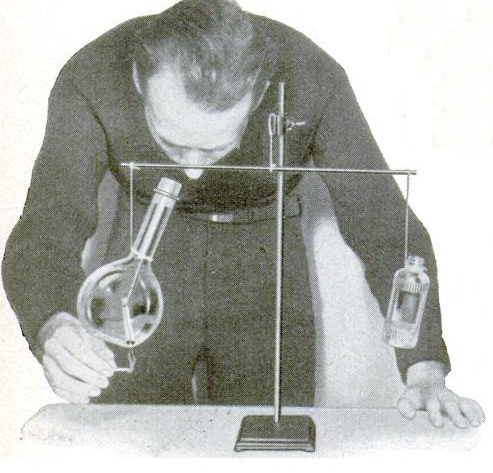
This little experiment, from 80 years ago, takes into account the amount of oxygen, and proves that the total mass doesn’t change during the combustion process. You do this by placing some matches inside a sealed glass flask. You carefully place it on a balance. Since the matches are sealed inside, you’re not able to strike them. To ignite them, you heat up the outside of the glass. Eventually, the matches will burst into flame and burn until all of the oxygen in the flask is consumed.
The balance won’t move, since the weight inside the container remains exactly the same. The weight of the matches plus the oxygen will exactly equal the weight of the burnt matches plus the weight of the smoke. Since it’s all sealed inside the same container, that weight won’t change.
If you wanted, you could take it a step further and repeat the experiment with the flask open. In this case, the matches would burn longer. Oxygen would be able to go in, and the smoke would be able to go out. Therefore, the weight would change. Would it go up or down?
You can easily adapt this idea to your next science fair assignment with a hypothesis along the lines of, “mass is conserved during combustion.” While that other kid is busy fumbling with the paper mache volcano, the teacher will be suitably impressed that you’re smarter than Sir Walter Raleigh, and you’ll undoubtedly go home with the first prize.
The photo and experiment appeared in the August 1937 issue of Popular Science.

Pingback: Science Fair Ideas: Fun With Air Pressure (With Optional Explosive Gas) | OneTubeRadio.com